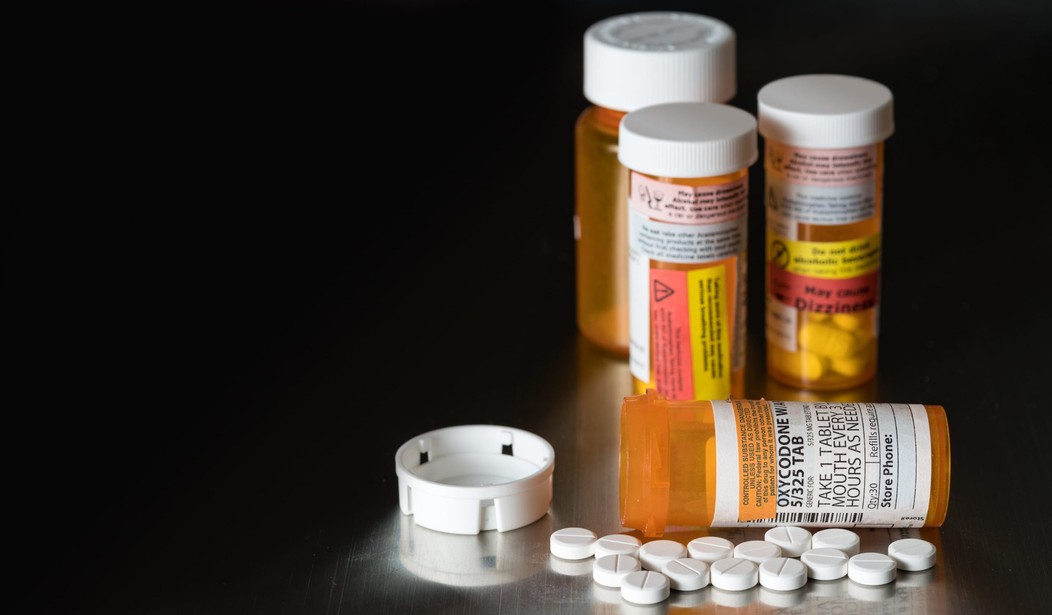
WASHINGTON – Sen. Ed Markey (D-Mass.) said building a border wall on the U.S.-Mexico border is a “distraction” from the opioid crisis, arguing that money allotted for the project would be better spent on opioid treatment.
President Trump has said a wall on the U.S.-Mexico border would help combat the flow of illegal drugs such as heroin into the United States.
“A Trump administration will secure and defend our borders. And yes, we will build a wall,” Trump said during the presidential campaign in a speech about the opioid epidemic. “A wall will not only keep out dangerous cartels and criminals, but it will also keep out the drugs and heroin poisoning our youth.”
According to the National Institute on Drug Abuse, “research suggests that misuse of these drugs may open the door to heroin use. Nearly 80 percent of Americans using heroin (including those in treatment) reported misusing prescription opioids prior to using heroin.”
PJM asked Markey if he agreed with Trump’s view that a border wall would serve as an effective tool to stop drug trafficking and the opioid epidemic.
“The wall is a distraction. We don’t need a wall. We need treatment. We don’t need to find $30 billion to build a wall. We need to release $500 million to states to institute prevention, treatment and recovery programs. It would be great if we could commit $30 billion to treatment programs, to programs that really do go to the heart of the program,” Markey said during a conference call on Tuesday about Dr. Scott Gottlieb, Trump’s nominee to lead the Food and Drug Administration (FDA).
“But I think the president is going to continue to go down that track and I don’t think it’s incidental that he wants to have that $30 billion while simultaneously cutting the [Health and Human Services] budget by 18 percent. You need to pay for that wall, and unfortunately that payment is going to come of the programs we really do need,” he added.
Markey argued that Gottlieb could not effectively address the opioid crisis due to his ties with the pharmaceutical industry. Gottlieb was deputy FDA commissioner under President George W. Bush’s administration and has worked as a consultant for Bristol-Myers Squibb and GlaxoSmithKline.
Markey said Gottlieb believes drug safety does not need “strong” oversight.
“We are suffering this public health epidemic because big pharma pushed pills they knew were dangerous and addictive, the FDA approved them, often without expert counsel, and doctors, because they do not have mandatory education on these drugs, prescribed them,” he said. “It is a vicious and deadly cycle that has turned this nation into the United States of Oxy and it must stop.”
Markey said Dr. Gottlieb’s “big pharma formula is simple: take away DEA oversight over prescription opioids and give that authority to the FDA, then at the same time limit the FDA’s ability to utilize its full oversight authority over these addictive products.”
Sen. Sherrod Brown (D-Ohio) echoed Markey on Gottlieb’s nomination to run the FDA.
“Unfortunately, Dr. Gottlieb’s record indicates that as commissioner he wouldn’t take the epidemic and the FDA’s authority to rein in prescription painkillers and other drugs seriously enough, which is why I will oppose his nomination,” Brown said. “Dr. Gottlieb has had a cozy relationship with big drug companies for decades.”
Brown argued that Gottlieb would be weak on oversight of the tobacco industry, specifically when it comes to the safety of e-cigarettes.
“We need a strong public health advocate who will continue to stand up to big tobacco the way that Senator Markey has,” he said.
Gottlieb’s confirmation hearing before the Senate Health, Education, Labor and Pensions Committee was Wednesday.
Chairman Lamar Alexander (R-Tenn.) said the nominee comes “with impressive qualifications from nearly every perspective.”
“You were a practicing physician and hospitalist for many years. You received your medical degree at Mount Sinai School of Medicine and completing your residency in internal medicine at the Mount Sinai Hospital. You have held three positions in the Department of Health and Human Services, including two positions at the FDA as deputy commissioner from 2005 to 2007 and before that in 2003 to 2004 as a senior advisor to Commissioner Mark McClellan, and as the FDA’s director of Medical Policy Development,” Alexander said.
“Since your time in government, you have studied health policy as a Resident Fellow at the American Enterprise Institute. You are also a prolific writer and speaker on medical innovation, and no stranger to testifying before Congress. You have testified in front of Congress 18 times on a variety of issues including the drug approval process, drug costs, drug shortages, importation, and healthcare reform,” the chairman added. “You are also a cancer survivor. You know firsthand how medical treatments affect patients and their families.”







Join the conversation as a VIP Member